KARASHIELD™
Clinically Tested & Patented Herbal Immune Support Formula –Produces Results Beginning in Just 7 Days*
Why Choose KaraShield™
Produces noticeable immune function improvements within 7 days*
Fortifies the immune system for optimal defense against environmental & seasonal challenges*
Boosts immune function*
Supports normal C-reactive protein levels*
Keeps you healthy against immune system challenges*
Supports a healthy inflammatory response*
Supports a healthy immune system*
Supports optimal well-being & immune health*
Safe & highly effective vegan formula*
Non-GMO Project Verified, Kosher & HALAL*
Available in powdered form for formulating flexibility*
Can be added to supplements, foods, beverages and more*
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
Disclaimer: This website summarizes the results of a randomized, double-blind, parallel and placebo-controlled study. KaraShield™ is not offered for sale to consumers. The information contained herein is only intended for use by manufacturers for product development purposes and should be reviewed by their legal counsel before the information is used on labels, in marketing or promotional materials or as support for any claim made in connection with a product that contains KaraShield™.
3-IN-1 SCIENTIFICALLY PROVEN BENEFITS FOR:
1 – IMMUNE SYMPTOMS*
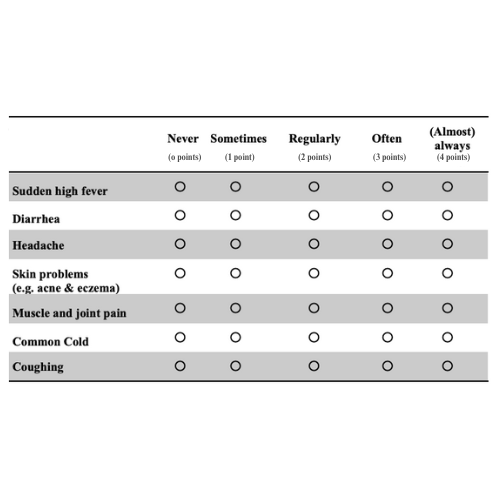
81.5% decrease in upper respiratory tract symptoms vs 39% for placebo* [Clinically Documented]
53.5% decrease in subjects assessment of frequency of symptoms vs 9.1% for placebo* [Immune Status Questionnaire]
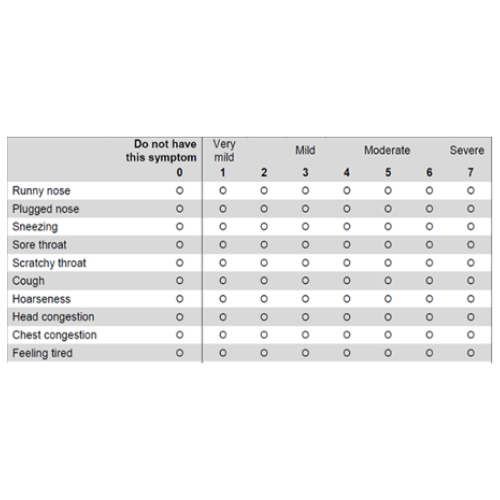
82.5% decrease in severity of upper respiratory tract symptoms vs 34.5% for placebo* [WURSS-24 Symptom Score]
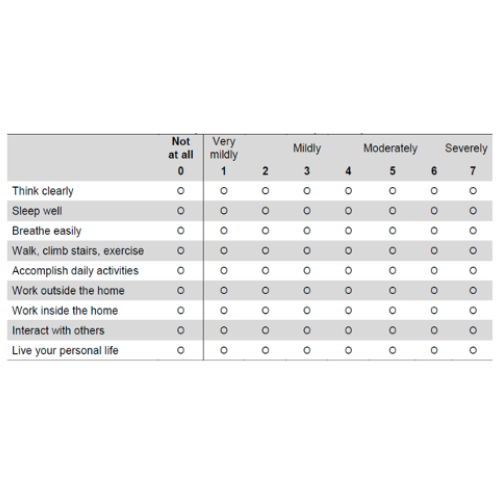
60.8% improvement in ability to do daily functions vs 33.4% for placebo* [WURSS-24 Function Impairments & Abilities Score]
35.1% increase in overall feelings of wellness vs 0.6% increase for placebo* [WURSS-24 Global Severity & Global Change Score]
2 – IMMUNE BOOST*
12.3% increase in serum Immunoglobulin G (IgG) vs 4.7% decrease for placebo* [Blood Markers of Immune Function]
3 – HEALTHY INFLAMMATORY RESPONSE*
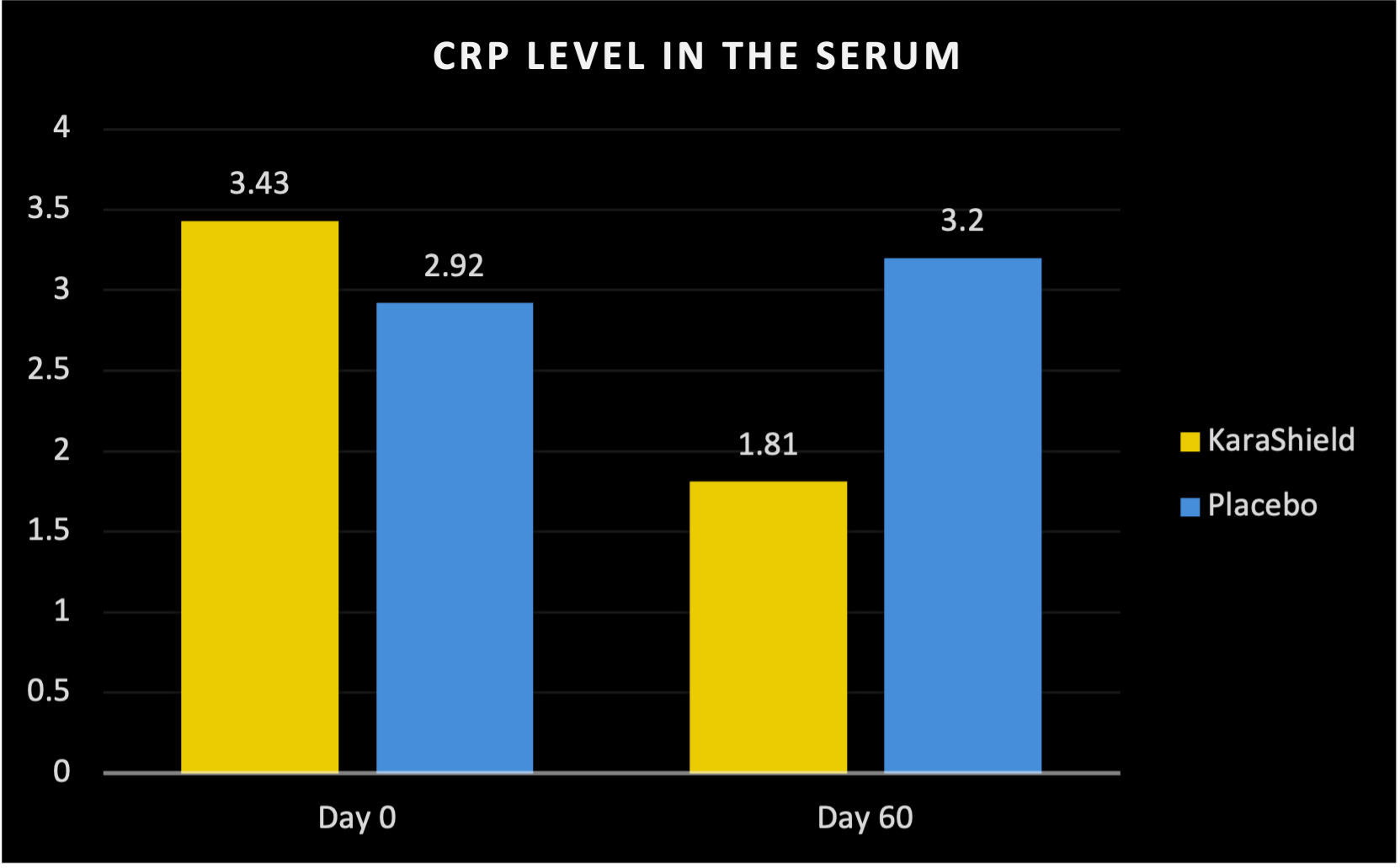
47.2% decrease in C-reactive protein vs 9.6% increase for placebo* [Blood Markers of Immune Function]
THE CLINICAL STUDY
-

OBJECTIVE
To study KaraShield’s ability to support immune function.
-

PARTICIPANTS
120 healthy subjects who experienced more than 2 clinically confirmed upper respiratory symptoms in the past 2 months – split evenly between two groups.
-

METHOD
A randomized, double-blind, parallel, placebo-controlled study with measurements at Day 7, 15, 30 and 60.
-

DAILY DOSAGE
500 mg of KaraShield for one group and 500 mg placebo for the other group.
CLINICALLY VALIDATED RESULTS
KaraShield herbal formula is clinically validated and proven to improve immune function through 3 independent variables:
Clinically documented number of upper respiratory tract immune symptoms
Subjective questionnaires
Improvements in serum markers of immune function
Participants not only reported noticeable improvements in how they felt, but also blood markers improved as expected.*
Scientifically Proven Benefits for Immune Symptoms*
81.5% Decrease in Upper Respiratory Tract Symptoms vs 39% for Placebo*
Clinically Documented Upper Respiratory Tract Symptoms
When comparing the 60 day period before the study to the 60 day period from the start to the end of the study, KaraShield subjects experienced a statistically significant 81.5% decrease in upper respiratory tract related immune symptoms compared to a 39% decrease for the placebo group.*
Clinically Documented Upper Respiratory Tract Symptoms Include: Common cold, cough, sore (scratchy) throat, nasal discharge (runny nose), nasal obstruction (plugged or congested), sneezing, headache, tiredness/body ache, chillness, etc. due to the common cold and/or seasonal change-related symptoms (except the allergic conditions)
53.5% Decrease in Subjects Assessment of Frequency of Symptoms vs 9.1% for Placebo*
Immune Status Questionnaire (ISQ)
KaraShield subjects experienced a statistically significant improvement in this immunity status score starting at day 15. By Day 60, KaraShield subjects experienced a 53.5% decrease in immune symptoms compared to a 9.1% decrease for the placebo.*
82.5% Decrease in Severity of Upper Respiratory Tract Symptoms vs 34.5% for Placebo*
Wisconsin Upper Respiratory Symptom Survey (WURSS-24 Symptom Score) - A Measure of the Severity of the Symptoms
KaraShield subjects experienced a statistically significant decrease in the severity of immune symptoms starting at day 7. By Day 60, KaraShield subjects saw an 82.5% decrease in the severity of the symptoms compared to a 34.5% decrease in the placebo group.*
60.8% Decrease in Functional Impairments vs 33.4% for Placebo*
Wisconsin Upper Respiratory Symptom Survey — Functional Impairments & Abilities Score (WURSS-24 Functional Impairments & Abilities Score) - A Measure of the Difficulty Subjects Experienced in Doing Different Daily Functions
KaraShield subjects experienced a statistically significant decrease in functional impairments starting at day 7. After 60 days, KaraShield participants had a 60.8% decrease in functional impairments, compared to only a 33.4% decrease in the placebo group.*
35.1% Increase in Overall Feelings of Wellness vs 0.6% Increase for Placebo*
Wisconsin Upper Respiratory Symptom Survey Global Severity & Global Change Score - A Measure of the Subjects’ Overall Well Being
After 60 days, KaraShield participants had a 35.1% increase in overall feelings of wellness compared to a 0.6% increase in the placebo group.*
Scientifically Proven Benefits for an Immune Boost*
12.3% Increase in Serum IgG vs 4.7% Decrease Placebo*
Serum IgG - Antibodies
KaraShield subjects experienced a 12.3% increase in Serum IgG compared to a 4.7% decrease in the placebo group at the end of the 60 day study.*
Scientifically Proven Benefits for a Healthy Inflammatory Response*
47.2% Decrease in C-reactive Protein vs 9.6% Increase Placebo*
C-Reactive Protein - A Measure of Inflammation
KaraShield subjects experienced a 47.2% decrease in C-reactive Protein compared to a 9.6% increase in the placebo group at the end of the 60 day study.*
THE HERBAL FORMULA
KaraShield is a blend of these herbal extract ingredients with a specific unique blending ratio and standardized to specific active compounds:
-

Green Chiretta
Andrographis Paniculata
-

Ashwagandha
Withania Somnifera
-

Moringa
Moringa Oleifera
-

Holy Basil
Ocimum Sanctum
OUR QUALITY STANDARDS
Karallief® has successfully united the powerful forces of nature, science and innovation to develop our breakthrough KaraShield formula.
Our company is unlike many ingredient suppliers that use inferior, untested materials that lack quality control measures. Instead, we pride ourselves on offering safe, efficacious and scientifically backed ingredients that are tested through thorough quality control procedures using high-end testing equipment.
Rigorous Quality Control
The testing performed covers a wide range of categories, including but not limited to: Physical parameters, chemical parameters, HPTLC chromatographic fingerprinting, HPLC quantified active compounds, impurity profile, microbial profile, residual solvents, residual pesticides, heavy metals profile and aflatoxins. The stability of every ingredient is periodically monitored through the product's lifetime and elaborate documentation is maintained to ensure traceability at all levels.
Comprehensive Quality Assurance
KaraShield is manufactured in ISO 22000 and GMP certified facilities. There are specific HACCP and HARPC procedures that are followed to analyze each step in the production process. This enables us to assess the risk of any potential biological, chemical or physical hazard and contamination, and if one is identified, we implement specific control measures to address the issue, ensuring the product is manufactured safely, and free of hazards and cross-contamination.
Control Samples & Stability Testing
Control samples for every single batch of KaraShield is maintained to facilitate traceability and to ensure that it consistently meets our high-quality standards. Stability testing of the product, which includes real-time and accelerated stability testing, is also performed at specified time intervals. By performing these tests, it helps us determine the shelf life of our products and monitor the characteristics of the product over time to ensure continued efficacy and purity.
Command the Attention Your Brand Deserves!
Create a Product with KaraShield™
CONTACT US TO LEARN HOW WE CAN WORK TOGETHER
Karallief® Inc.
Email: KaraShield@Karallief.com
Address: 75 Arlington St. Suite 500, Boston, MA 02116, USA
Scientific References
Lichtenberger F, Rajendran K, Layton K, Lakshminarayan S, Krishnamurthy L, Deep DK. Assessment of KaraShield™ properties in supporting the immune health of healthy subjects: a randomized, parallel, double-blind, placebo-controlled clinical study. Int JBasic Clin Pharmacol 2024;13:1-12.
Moyad M, Robinson L, Zawada E Jr, Kittelsrud J, Chen D, Reeves S, Weaver S. Effects of a modified yeast supplement on cold/flu symptoms. Urol Nurs 2008 Feb;28(1):50-5.
McFarlin B, Carpenter K, Davidson T, McFarlin M. Baker's Yeast Beta Glucan Supplementation Increases Salivary IgA and Decreases Cold/Flu Symptomatic Days After Intense Exercise. Journal of Dietary Supplements, 10(3):171–183, 2013.
Fuller R, Moore M, Lewith G, Stuart B, Ormiston R, Fisk H, Noakes P, Calder P. Yeast-derived β-1,3/1,6 glucan, upper respiratory tract infection and innate immunity in older adults. Nutrition Volumes 39–40, July–August 2017, Pages 30-35.
Zakay-Rones Z, Thom E, Wollan T, Wadstein J. Randomized Study of the Efficacy and Safety of Oral Elderberry Extract in the Treatment of Influenza A and B Virus Infections. The Journal of International Medical Research 2004; 32: 132 – 140.
Zakay-Rones Z, Varsano N, Zlotnik M, Manor O, Regev L, Schlesinger M, Mumcuoglu M. Inhibition of Several Strains of Influenza Virus in Vitro and Reduction of Symptoms by an Elderberry Extract (Sambucus nigra L.) during an Outbreak of Influenza B Panama. The Journal of Alternative and Complementary MedicineVol. 1, No. 4
KARALLIEF® INC. 75 Arlington St. Suite 500, Boston, MA 02116, USA | KaraShield@Karallief.com
© Copyright 2024
Privacy Policy
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
This website summarizes the results of a randomized, double-blind, parallel and placebo-controlled study. KaraShield™ is not offered for sale to consumers. The information contained herein is only intended for use by manufacturers for product development purposes and should be reviewed by their legal counsel before the information is used on labels, in marketing or promotional materials or as support for any claim made in connection with a product that contains KaraShield™.